An ongoing dialogue on HIV/AIDS, infectious diseases,
May 2nd, 2013
How to Interpret Medical Breakthroughs in the Mainstream Media
There it is, right in your daily paper, on your tablet or computer screen, or wherever you get your news today — a headline about a great medical breakthrough everyone’s been waiting for:
Scientists on brink of HIV cure
Researchers believe that there will be a breakthrough in finding a cure for HIV “within months”
Yes, I read this exact headline recently. Here’s the full article, published in the English newspaper the Daily Telegraph. It details how some Danish researchers have figured out a way for “the HIV virus to be stripped from human DNA and destroyed permanently by the immune system.”
Furthermore, they are “expecting results that will show that finding a mass-distributable and affordable cure to HIV is possible.”
 By all means, go ahead and read the full piece; you’ve got 20 free reads on the Telegraph website. As a treat, there’s a colorful stock photo too, showing red blood cells floating through some blood vessels, along with a few HIV virions glowing bright green — it’s very Fantastic Voyage-esque, minus Raquel Welch in her scuba gear.
By all means, go ahead and read the full piece; you’ve got 20 free reads on the Telegraph website. As a treat, there’s a colorful stock photo too, showing red blood cells floating through some blood vessels, along with a few HIV virions glowing bright green — it’s very Fantastic Voyage-esque, minus Raquel Welch in her scuba gear.
But return here for a moment, please. I’m going to recommend three simple steps to getting the most from this — and other medical breakthroughs — in the mainstream media.
Step 1: Be a skeptic. As exciting as curing HIV would be, and no matter how much you’d like this to happen, just think for a moment about the plausibility of this story. Are scientists really on the “brink” of curing HIV? If so, why is this only appearing in the U.K. Telegraph? Trust me, this brink-of-cure has not yet appeared in peer-reviewed medical journals or at scientific meetings. And wouldn’t you expect this kind of advance, if real, to show up everywhere in media land? Fire up that Google machine, and see what you can find about it elsewhere — lo, it’s the great following herd, all stampeding after that same U.K. Telegraph story. And importantly, here’s a New York Times piece on the very same general subject — HIV cure — and they don’t even mention these Danish researchers. Sure, the Times misses some stories, but it’s got some pretty impressive Health and Science sections — could they miss this, researchers ON THE BRINK OF CURING HIV, no less? I think not. So perhaps Mr. U.K. Telegraph Science Reporter is exaggerating a bit, for the sake of his story, of course.
Step 2: Don’t be a complete snob — give the story a chance. This is the other side of that same coin. Sure, it’s been a challenge curing HIV, but we’ve got that Berlin patient (now living in Las Vegas, by the way) — he’s cured. And the baby from Mississippi, he/she is cured (sort of). Plus, a whole army of smart HIV researchers actively tackling the problem as we speak. In fact, this very same approach cited by the Danish researchers — stimulating the HIV reservoir with an HDAC inhibitor — is a leading candidate for a viable cure strategy; it’s being looked at by many groups. Hey, why can’t Good Ol’ Ole Søgaard and his team be the first to succeed? The extra funding provided by the Danish government — 12 million Danish kroner! — is further evidence of the soundness of Professor Søgaard’s approach.
Step 3: After all that, land someplace between Steps 1 and 2. Take a deep breath. Read the full piece.
Discard the fluff: Brink of cure; 12 million Danish kroner; expect a cure to be available in months; you can distribute it to millions; it’s affordable, too.
Focus on the facts: Some Danish researchers have some funding to investigate a potentially promising HIV cure strategy; they are testing it in a small number of people; some European scientists may soon be collaborating; we have no actual results yet to report.
After these three steps, all these medical breakthroughs — on HIV, cancer, Alzheimer’s, weight loss, male-pattern baldness, you-name-it — make a lot more sense.
Even if they are less exciting. Enjoy the movie.
[youtube https://www.youtube.com/watch?v=0Sp2bJg_wqI&w=560&h=315]
April 29th, 2013
Data Safety Monitoring Board Closes HIV Vaccine Study — the End of Adenovirus as a Vaccine Vector?
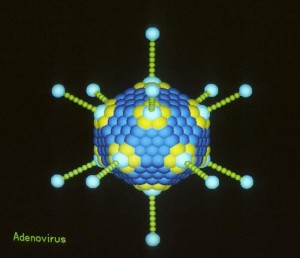 On Friday, the NIH announced that HVTN 505, a clinical trial of an HIV vaccine using an adenovirus vector, would be stopped based on a finding of futility by an independent DSMB.
On Friday, the NIH announced that HVTN 505, a clinical trial of an HIV vaccine using an adenovirus vector, would be stopped based on a finding of futility by an independent DSMB.
The study had enrolled some 2500 high-risk gay men in the United States. Here are the three key findings leading to this action:
- New infections in placebo arm: 30
- New infections in active vaccine arm: 41
- Among those who got infected, no effect of the vaccine in lowering HIV viral load compared with placebo
While the difference between the two is numerically but not statistically different, it’s clearly in the wrong direction.
Furthermore, it hearkens back to the results of the STEP study — another HIV vaccine trial using an adenovirus vector, Ad5 — that demonstrated an increased susceptibility to HIV infection among participants with pre-study immunity to adenovirus. Though HVTN 505 excluded those who had antibodies to Ad5, the results from HVTN 505 suggest that something else might be going on, something related to Ad5 and protective HIV immunity.
(Confession: I have no idea what that might be.)
As with all studies closed by DSMBs, the detailed data analysis starts in earnest now. And though this outcome is clearly disappointing, it will be fascinating to see what the full results show, and most importantly whether we can learn anything about a way forward in this challenging research area.
April 25th, 2013
In Praise of the Diversity of Being an Infectious Diseases Specialist
 I have a friend who is a thoracic surgeon.
I have a friend who is a thoracic surgeon.
Imagine his daily agenda:
5:00 AM: Oops, slept late.
5:30 AM: Drive to hospital; not much traffic.
6:00 AM: Round on inpatients. They’re all doing great! Wonder why they’re not more talkative.
6:15 AM: Coffee, gossip with other surgeons.
7:00 AM: Get in scrubs, prepare for first case.
7:15 AM: First case. Remove this person’s esophageal tumor.
11:00 AM: That was fun! Another person saved.
11:15 AM: Next case — bronchoscopy/mediastinoscopy; darn, non-small-cell lung cancer. Cigarettes are bad.
1:00 PM: Time to get lunch.
1:10 PM: Finish grant proposal. Give feedback to army of office staff, nurses, and PAs, who work for us.
2:00 PM: Next case. Remove this person’s lung nodule.
4:00 PM: Granulomas on frozen section? Great, it’s not cancer! Call ID consult.
5:30 PM: Start new grant proposal
7:00 PM: Time to head home. I wonder what part of the chest I get to remove tomorrow?
Now I don’t really know what his day is like. He might as well be piloting a hovercraft in the Everglades, periodically pausing to shoot crocodiles — that’s about how close the ID doctors’ daily life is to this imagined Thoracic Surgery life.
But I was thinking about this the other day when I was talking to him during a neighborhood party, because he implied mystification about what we do. In fact, he quickly acknowledged he couldn’t do what we do in a million years. “You guys know everything,” he said.
(Thank you. I didn’t disabuse him of this view.)
And while we might not — okay, don’t — know everything, it’s true we get to see absolutely everything that’s going on in the hospital. It really is quite extraordinary how diverse the cases we ID doctors see. On rounds or during clinic recently, I saw or heard about individual patients with the following medical/surgical/social/miscellaneous histories:
- Shot a raccoon.
- Weight > 700 pounds. [Unable to verify exactly.]
- Weight = 83 pounds.
- Adult with complex congenital heart disease.
- Six total joint replacements — of the same joint.
- Just arrived here from Mali.
- Has a cat, two dogs, two birds, a guinea pig, and a rabbit. [All completely unrelated to his ID problem, but interesting nonetheless.]
- Retired New Orleans chef, now sells pickles from his van.
- CD4 = 6.
- CD4 = 660.
- Reported vodka consumption: 1-2 liters/day. [They say always double what people report.]
- Fever > 107 Fahrenheit.
- Tattoos on her ears — just her ears.
- Started smoking at age 11 in Venezuela.
- 80% surface area burn.
- Speaks only Hmong.
- Sells real estate on the Baja Peninsula, lives in Boston.
- Regular Tough Mudder participant.
Note I deliberately left out specifically why they needed ID consults, but you can probably imagine in most of the cases. (Maybe not the pickle guy.)
But ID issues aside, isn’t it incredible what we learn about medicine — and people — just from doing our job? What a privilege.
Almost makes me regret whining about what we get paid.
April 23rd, 2013
Two Papers, Four Sofosbuvir Studies, and Soon the End of “Interferonologists”
 Today, as the The International Liver Congress is about to start, two papers are published in the New England Journal of Medicine on sofosbuvir, the investigational anti-HCV nucleotide submitted to the FDA for approval earlier this month.
Today, as the The International Liver Congress is about to start, two papers are published in the New England Journal of Medicine on sofosbuvir, the investigational anti-HCV nucleotide submitted to the FDA for approval earlier this month.
Each paper actually includes within them two studies. (For some reason, all the studies sound like 1950s science fiction magazines.) Typical of the frenetic pace of drug development for HCV, the trial designs are a byzantine mish-mash of study populations, strategies, and endpoints, which can make interpretation complex. As a result, here’s an attempt to summarize some key outcomes, with a focus on the bottom line — cure — making the not-so-bold leap that “sustained virologic response at 12 weeks” = “cure”.
One paper looked at treatment-naive patients with any HCV genotype, and included the NEUTRINO and FISSION studies. NEUTRINO was a single-arm treatment with peg IF, RBV and sofosbuvir for genotypes 1, 4, 5, and 6, and FISSION a randomized comparison between sofosbuvir and interferon (both with RBV) for genotypes 2 and 3:
- NEUTRINO: Genotype 1, 12 weeks of peg IF + RBV + sofosbuvir: 89% cure
- NEUTRINO: Genotype 4, 12 weeks of peg IF + RBV + sofosbuvir: 96% cure (Note: very few genotype 5 or 6 patients)
- FISSION: Genotype 2, 12 weeks of RBV + sofosbuvir: 97% cure
- FISSION: Genotype 3, 12 weeks of RBV + sofosbuvir: 56% cure
In the randomized comparison for the genotype 2/3 patients, response rates were similar between sofosbuvir for 12 weeks and interferon for 24 weeks (both with RBV); the sofosbuvir strategy was better tolerated. No resistance to sofosbuvir was detected in any patient.
The other paper — remember, also with two studies — looked only at patients with genotype 2 or 3 HCV, and in whom “peginterferon is not an option”. In one study (POSITRON), this could be for multiple reasons, such as prior adverse effects, concurrent medical conditions, or just refusing interferon; they were randomized to RBV + either sofosbuvir or placebo for 12 weeks. In the other study (FUSION), these were patients who did not respond to prior treatment with interferon, randomized this time to 12 or 16 weeks of RBV + sofosbuvir. Again, some cure rates (all genotype 2 or 3):
- POSITRON: Interferon not an option, 12 weeks RBV + sofosbuvir: 78% cure
- POSITRON: Interferon not an option, 12 weeks RBV + placebo: 0% cure (Note: must have included this for safety reasons, right?)
- FUSION: Prior non-response to interferon, 12 weeks RBV + sofosbuvir: 50% cure
- FUSION: Prior non-response to interferon, 16 weeks RBV + sofosbuvir: 73% cure
Patients with cirrhosis and/or genotype 3 didn’t do as well — extending the treatment to 16 weeks seemed to increase cure rates
No doubt these are terrific results, suggesting that sofosbuvir-based therapy will be both more effective and better tolerated than current standard of care. And also no doubt this week’s “Liver Congress” will have further interferon-sparing studies for HCV that could make these published studies seem out of date, even before the sofosbuvir is approved.
Nonetheless, as of right now — April 23, 2013, 2:43 PM EST — as an editorialist appropriately notes, it’s not quite time to say farewell to the “Interferonologists” among us who spend the bulk of HCV treatment time managing side effects.
The good news is that time is undoubtedly coming soon.
April 20th, 2013
Postexposure Prophylaxis (PEP) After Blast Injuries
From a colleague came this query:
We are being consulted by surgeons who are finding within blast victims tissues from other humans. We have been offering post-exposure prophylaxis. Have you folks developed any policies re PEP for explosion victims?
Welcome your thoughts,
P
Needless to say, the bombing victims are currently facing far greater challenges than exposure to blood-borne pathogens. But it’s a very reasonable query and, as it turns out, there’s an existing guideline that addresses the issue, published in MMWR in 2008.
Entitled “Recommendations for Postexposure Interventions to Prevent Infection with Hepatitis B Virus, Hepatitis C Virus, or Human Immunodeficiency Virus, and Tetanus in Persons Wounded During Bombings and Other Mass-Casualty Events” — MMWR has a real knack for titles — it’s a valuable resource for a a very tricky clinical problem. It appropriately starts by stating that “Decisions regarding the administration of prophylaxis after a mass-casualty event are complex,” then goes on to make the the following recommendations:
So in general, post-exposure prophylaxis for HIV is not recommended after bombings or other mass casualty events.
Still, the fine print does say it might be recommended in certain situations. One could imagine blast injuries in very high prevalence settings (Sub-Saharan Africa, certain urban areas in the USA) meeting this threshold.
Remember also that these were published in 2008, and at that time one major factor in the decision to withhold PEP was the toxicity of the intervention, which was zidovudine/lamivudine plus lopinavr/ritonavir. Today, with TDF/FTC and raltegravir the new standard of care, PEP is far better tolerated. (See here for the excellent New York PEP guidelines.)
So the bottom line? In grisly settings such as the one described in the above email — tissue of other humans embedded into humans — giving PEP is certainly defensible, guidelines notwithstanding.
April 10th, 2013
Simeprevir and Sofosbuvir Submitted to FDA — Clock Ticking on Boceprevir, Telaprevir, Even Interferon
 Two weeks, two companies, two press releases, two future HCV drugs that begin with “S”:
Two weeks, two companies, two press releases, two future HCV drugs that begin with “S”:
- March 28, 2013: Janssen Research & Development announced that it has submitted a New Drug Application to the FDA seeking approval for simeprevir (TMC435), an investigational NS3/4A protease inhibitor, administered as a 150 mg capsule once daily with pegylated interferon and ribavirin for the treatment of genotype 1 chronic HCV in adult patients.
- April 8, 2013: Gilead Sciences announced that it has submitted a New Drug Application to the FDA for approval of sofosbuvir, a once-daily oral nucleotide analogue for the treatment of chronic HCV infection. The data submitted in this NDA support the use of sofosbuvir and ribavirin as an all-oral therapy for patients with genotype 2 and 3 HCV infection, and for sofosbuvir in combination with ribavirin and pegylated interferon for treatment-naïve patients with genotype 1, 4, 5 and 6 HCV infection.
For several years now, all clinicians who see patients with hepatitis C have been promising them better treatments “soon”, with admittedly little precision about exactly what this “soon” actually means.
But with these filings, we have a pretty good idea. It will be less than a year — maybe much less. Why is that?
- The FDA has roughly 10 months to review these applications. (The exact timing is somewhere on the FDA web site, see if you can find it.)
- These two drugs have looked pretty great in clinical studies to date.
- The bar to leap over to be substantially better than current standard -of-care treatment for HCV isn’t exactly high — a fact that could get at least one drug, if not both, priority review and even more rapid approval.
- One of my patients has joked that I’ve been saying “in a few years” for availability of better HCV treatment options for at least “a few years” now — so my time is up.
Of course, stuff could happen that holds up the approvals. A toxicity could arise that hasn’t been reported previously, or a tricky drug-drug interaction could crop up. Or the sun could explode.
But if these things don’t happen, then for patients with HCV genotypes 2 and 3, a non-interferon option looks like it’s right around the corner — sofosbuvir and ribavirin. For those with genotypes 1 and 4, a shortened, more effective, and all-round improved interferon-based regimen will arrive at the same time — pegylated interferon/ribavirin plus either simeprevir or sofosbuvir.
And even better, how about an off-label combination of these two new drugs as in this clinical study — in which case the interferon can be dropped entirely, right?
Let the choir sing out when that happens, and enjoy a hearty celebratory brunch at the famous Cambridge deli pictured above in honor of the new regimen.
April 5th, 2013
Another “Important Advance” in HIV Vaccine Research?
 On reading this other real news about a single patient and how it may shape the future of HIV vaccine research, I decided to write the following fake news, drawing liberally on many similar stories over the years:
On reading this other real news about a single patient and how it may shape the future of HIV vaccine research, I decided to write the following fake news, drawing liberally on many similar stories over the years:
Scientists today reported a discovery that could finally pave the way for an effective AIDS vaccine. In the study, published in the journal Science, they describe a single person who lacks evidence of HIV despite extensive testing using even the most sensitive tests.
Gustav Blinkerhood, a research scientist from the University of Minnesota and the lead author on the paper, was quick to caution that the patient may have tested negative for HIV simply because he doesn’t have it, and never did.
“We cannot find any traces of the virus,” said Blinkerhood about the case. “All the tests are negative.”
Not only was there no direct evidence of HIV — the virus that causes AIDS — the patient also lacked the tell-tale antibodies that are present in someone exposed to HIV who has acquired it.
Blinkerhood’s research team plans further testing of other people who don’t have HIV. “Our hypothesis is that they will test negative as well,” he said.
Researchers have been stymied for years in their efforts to develop an effective HIV vaccine because the virus changes so darn much, evading the immune system. It is hoped that this study of uninfected people who test negative for the virus will spearhead a new round innovative research.
Dr. Anthony Fauci*, the Director of the National Institute of Allergy and Infectious Diseases, praised the scientific rigor of study yet also stated that the development of an AIDS vaccine remains a major challenge.
“In is important that I comment on all widely publicized HIV research in this way,” said Dr. Fauci. “And that’s what I’m doing right here.”
Dr. Jane Greezley, a Professor of Microbiology at Tufts University, wrote an accompanying editorial in the journal. She agreed with Fauci, adding “I was a reviewer of this paper, and recommended it be accepted for publication, then the editors asked me to write this commentary — maybe they’ll accept my next paper in return.”
The patient, who is entirely healthy and has requested anonymity, is named Charles Gallagher. He lives in Minneapolis at 17 Fairfield Road, and his cell phone number is 292-344-8664.
(*Hey, if I had a big HIV news story, I’d call him too!)
Yes, this should have been posted on April 1. But I was too excited by Infectious Diseases Exotica Day on Physician’s First Watch, sorry.
April 2nd, 2013
Banner Day for ID on Physician’s First Watch, and a Big Pitch to Sign Up Now
 Every weekday morning, right around the time the rest of my family gets up, the smart people at Physician’s First Watch send me an email listing the top medical news stories of the day.
Every weekday morning, right around the time the rest of my family gets up, the smart people at Physician’s First Watch send me an email listing the top medical news stories of the day.
Imagine my delight yesterday when the following were deemed worthy for specific mention:
- Coccidioidomycosis! Valley fever cases on the rise in the USA. You too can learn how to spell and pronounce it (only wimps say “cocci”), provided you practice, practice, practice…
- Mycobacterium abscessus!! Looks like person-to-person transmission can occur, at least according to this paper in the Lancet.
- Q fever!!! The Centers for Disease Control have issued guidelines on diagnosis and management of Coxiella burnetii (Q fever), thereby fulfilling what was undoubtedly a burning national need. (Sarcasm used with the utmost affection, CDC.)
Not only was I delighted by this threesome, but confess a bit surprised as well. After all, it’s not as if these are garden-variety ID problems; in fact, if all three of these diagnoses showed up in an ID case conference, it would be one heck of a meeting. By chance, one of these — M. abscessus — did get discussed at our conference this week, and boy was I prepared. Thank you, PFW.
All of which is a way of putting in a plug for Physician’s First Watch, which is concise, free of charge, and well worth the 5 seconds it takes to sign up for it.
Even when the coverage does not involve the World’s Greatest Medical Specialty.
March 28th, 2013
Poll: How Often Do You Measure CD4 Cell Counts?
Over in Clinical Infectious Diseases, a recent study pretty much nails the fact that routine measurement of CD4 cell counts in clinically stable patients is an all but useless exercise. As summarized by Abbie Zuger in Journal Watch, here’s the key finding:
When patients with an unrelated cause for an alteration in CD4-cell count such as severe infection, chemotherapy, or interferon treatment were excluded from the analysis, not a single patient in any group had a dip in CD4 count below 200 cells/mm3 after 2 years of continuous virologic control.
So I’ve been singing this tune for a while (large nose-enhancing video here), and as a result have been trying for some time to get my stable patients to reduce the frequency of CD4 monitoring — or even, as I note in this editorial, give it up entirely!
 And has this been a successful effort? For some patients, yes, but for others it’s hopeless — they simply can’t understand that this test, which was the cornerstone of HIV monitoring for decades, now provides us with information that has no role in determining what we do therapeutically (provided the viral load remains suppressed).
And has this been a successful effort? For some patients, yes, but for others it’s hopeless — they simply can’t understand that this test, which was the cornerstone of HIV monitoring for decades, now provides us with information that has no role in determining what we do therapeutically (provided the viral load remains suppressed).
In short, we order the test for emotional and sentimental reasons only — it’s reassuring to patients to hear that their CD4 cell count is stable, and we’ve been doing it for so long, why stop now. But are these good enough reasons? Before you answer in the affirmative, remember that unexplained drops in CD4 (which are not uncommon) have the opposite effect, and require frequent education about why the result won’t change our patients’ treatments.
So I ask you, providers of HIV care, the following burning question:
March 23rd, 2013
ID Doctors, Pets in the Medical History, and a Cute Puppy
 One of the things Infectious Disease doctors get teased about by our non-ID colleagues is our inclusion of pets in medical histories.
One of the things Infectious Disease doctors get teased about by our non-ID colleagues is our inclusion of pets in medical histories.
It’s part of the social history, where we list a grab bag of potential “exposures” that increase the risk of infection — where someone is from, what they do, plus travel, dietary practices, sex, drugs, alcohol, cigarettes, and PETS!
In our defense, to our critics I remind them that we take the best histories — if you don’t believe me, read this — but must acknowledge that the pet thing is not always relevant. Ok, it barely ever is relevant. But it sure is fun.
In order to examine this issue more closely, I offer the following anecdotes and observations — none of them leading to brilliant pet-related diagnoses — taken from real life clinical practice:
- I once cared for someone who was very funny — so much so that he was funny for a living. When I asked him if he had any pets, he told me he had 15 cats. How did he do that? His response: “Easy — when you have 14 cats, you get one more, and if you have 16, one of them dies or runs away.” (FYI, I think he stole this line.)
- A patient told me she was taking her dog’s deworming medicine for pinworm. She didn’t have pinworm, by the way, but these were her symptoms, so she was convinced. (Warning: video is kind of yucky.) The medication she was taking was something called “Panacur” (fenbendazole). While I like that name — Panacur sure sounds like it cures a lot of things — it’s only approved for use in sheep, cattle, horses, fish, dogs, cats, rabbits and seals.
- Dept. of Irony: Asked someone recently if he’d received his zoster vaccine, and he told me that he refused because the cost was way too high ($300), and not covered by insurance. Later, when I asked him about his pets, he told me he’d paid nearly $3000 for his dog’s various orthopedic ailments.
- It used to be that none of our patients from Haiti had pets. Now lots do. Is there a PhD anthropology thesis in there? I’d better ask Paul Farmer.
- Every so often, someone tells you he/she has a pet turtle, frog, or lizard. Should you warn them of the risk of salmonella, and ruin their fun? And if not, why ask? These are the kind of tough dilemmas ID doctors face every day.
- My first opportunity to witness the occasional absurdity of this pet obsession was during my fellowship. One of my attendings, an extremely detail-oriented type even by ID standards, was consulted by a trauma surgeon on a comatose man with fevers during a lengthy hospital stay after a motorcycle accident. She wrote in her note, “has parakeet named Fruitloop.” This surprised me, as the patient didn’t seem like the parakeet type. And I still wonder — how did my attending find this out? Here’s the dialogue that might have happened:
ID doctor: Does he have any pets at home?
Patient’s Mother (surprised at the question): Yes, he has a bird. We’re taking care of it since the accident.
ID doctor: What kind of bird is it?
Patient’s Mother (even more surprised): It’s a yellow parakeet.
ID doctor: What is the bird’s name?
Patient’s Mother: Fruitloop. (A pause.) Excuse me doctor, are you out of your mind?
So here’s a confession. This post is an excuse to show off a picture of our new puppy.
His name is Louie.


