An ongoing dialogue on HIV/AIDS, infectious diseases,
July 29th, 2013
CROI 2014: March 3-6, Boston, Massachusetts, USA, North America, Earth
Ok, that’s one mystery solved.
(We’ll do our best to have the ice and snow cleared away by then, but no promises — New England weather is the very definition of unpredictable.)
And now that we know the CROI 2014 dates, here’s some good news: I hear they’re working on getting the 2015 dates squared away as well.
If so, good work, new CROI organizers!
Keep an eye out for further details (deadlines, etc) here.
(Thanks to an un-named but very reliable source for this, who gave permission to share the info.)
July 23rd, 2013
Guess it Wasn’t the Acyclovir After All
Big news in baseball land is that superstar Ryan Braun has been suspended for the rest of the season for using performance enhancing drugs (PEDs).
And why is this information in an Infectious Diseases blog? Three main reasons:
- When Braun first got nabbed for PEDs in 2011, it was widely rumored it had something to do with treatment for herpes. Not surprisingly, I was confused about this, but reasoned that maybe — just maybe — it was due to an interaction between acyclovir and endogenous testosterone production. Pretty weak, I know.
- Braun then got the suspension dropped because the urine specimen wasn’t handled properly. In hindsight, Braun made some astounding comments after getting off, but no mention whatsoever of acyclovir.
- I will use any excuse imaginable to write about baseball.
Admittedly, the last one is probably the biggest factor here.
Ah, for a simpler, more innocent (!?) era …
July 20th, 2013
There’s No US News & World Report Infectious Diseases Ranking — Is That A Good Thing?
 As the internet burns through traditional print media, decimating anything paper in its path, several odd things have happened beyond simply putting those newspapers, magazines, and journals out of business. For example, Gourmet was subsumed into Bon Appetit — who could have predicted that? PLoS One and its ilk were born.
As the internet burns through traditional print media, decimating anything paper in its path, several odd things have happened beyond simply putting those newspapers, magazines, and journals out of business. For example, Gourmet was subsumed into Bon Appetit — who could have predicted that? PLoS One and its ilk were born.
And what about the hard-copy versions of the big three news magazines?
- Newsweek disappeared.
- Time has gone on quite a diet, getting thinner every week.
- US News & World Report, the awkwardly named and most serious of the three, initially decreased publication from weekly to biweekly, then to monthly, then stopped entirely in 2010.
That is, US News & World Report stopped publishing a news magazine — it still, of course, publishes the hugely popular rankings of colleges, grad schools, and hospitals, much to the delight (or chagrin) of administrators, marketers, and PR personel everywhere.
In many ways, USN&WR (can’t stand to write out the whole thing again, sorry) has morphed into a Michelin-like guide to the country’s “Best Anything” — take a look at this “Rankings” page — and who knows what they’ll rank next.
They already do high schools, law firms, nursing homes, summer vacations. How about museums, car washes, roller rinks, AM radio stations, salt and vinegar potato chips — the possibilities are endless. They could even rank news magazines, thought that would be a very short report.
As for the hospital rankings, which came out last week, I confess to having a pretty cynical view of the whole enterprise, despite the fact that our hospital does quite well.
(For the record, not this well, mind you, but not too bad. Marketers can’t resist this stuff.)
To explain my skepticism, let me ask some truly obvious questions which have anything but obvious answers: Isn’t measuring the quality of care a particularly tough challenge? What are the outcomes of interest? Survival? Complications? Patient satisfaction? Cost effectiveness? How is the care for the poor and/or uninsured, or the average Jane or Joe, or for the billionaire hedge fund manager or Saudi prince who can go anywhere? Which is more important, how a hospital does with particularly complex cases, or with “routine” care? How long is the average wait for an elevator? (We must have lost points on that one, trust me.)
In sum, who’s to say that the USWNR methodology is the right one?
It’s because of these and other concerns that I’m always relieved that Infectious Diseases is not one of the 16 specialties included in the rankings.
Or maybe I’m just worried that we’d come up short in their ID-specific criteria — our consults lack sufficient pet histories, we recommend too much indiscriminate carbapenem use, vancomycin levels are too low, some HIV patients have no documentation of their PPD status. In a best case scenario, we could use the results to improve areas of weakness, a kind of national quality assurance program that’s only slightly embarrassing.
Plus, wouldn’t listing ID in the specialties give our field some much-needed publicity?
What do you think?
July 14th, 2013
Will Dolutegravir Instantly Become the Integrase Inhibitor of Choice in Patients with Treatment Failure?
 Here’s the short answer : Yes. Probably.
Here’s the short answer : Yes. Probably.
And here’s why.
In a randomized, double blind clinical trial just published in the Lancet — it’s called SAILING — once-daily dolutegravir was compared to twice daily raltegravir in treatment-experienced patients. The site investigators could choose one or two other fully active agents to develop an optimized background regimen (OBR). There was a protocol-specified stratification based on fully-active darunavir and baseline viral load.
There were over 700 study subjects; they were recruited from 156 sites, located in every continent except Antarctica.
(Yes, 156 sites. Hey, it’s hard to find patients who fail treatment who are good candidates for clinical trials!)
And here are the results:
- Virologic outcome: Dolutegravir was superior to raltegravir, 71% suppressed at 48 weeks, vs 64%, p=0.03.
- Those experiencing virologic failure had less resistance on dolutegravir than raltegravir.
- In the hardest-to-treat subgroups — those with no active darunavir in their OBR, or the higher viral load stratum — the differences favoring dolutegravir over raltegravir were even greater than in study overall.
- Adverse events, serious or otherwise — pretty much the same.
Now I’ve said before that raltegravir is one of our very best antiretroviral agents: it’s highly potent, well tolerated, has very few drug-drug interactions, an extensive safety record dating back to clinical trials that started in the mid-2000s, and and even more extensive favorable clinical experience since its approval in 2007.
But dolutegravir is once daily, seems just as well tolerated as raltegravir, has a similar drug-drug interaction profile — and based in the results of SAILING, is probably more potent and less prone to resistance.
Note that elvitegravir isn’t currently in the discussion, since it’s not available except as part of this one pill — TDF/FTC/EVG/COBI — which can’t be given with boosted PIs, so is a difficult choice in those with treatment failure.
Bottom line: barring some unexpected toxicity when it gets into broader use after approval — always a possibility — dolutegravir will instantly become the integrase inhibitor (II, INI, INSTI, InSTI) of choice in treatment experienced patients with treatment failure.
(Still need a good abbreviation for this drug class.)
July 7th, 2013
Almost Annual Whine About No CROI Dates, and a New Temporary (I Hope) CROI Website
Believe or not, sometimes we know a year in advance the dates of the Conference on Retroviruses and Opportunistic Infections (CROI).
For example, we learned at the beginning of CROI 2010 that in 2011, it would take place February 27-March 3 — in Boston no less. Yay!
(The meeting was a bit shorter, but it did in fact start on Feb 27.)
But most of the time, “When is CROI next year?” is a free-floating mystery, one that can’t be solved until a surprisingly short time before the actual meeting.
We know that CROI will be in the winter, probably in February or March, and that abstracts will be due some time in the fall. Plus we know CROI will be held someplace in North America, though of course the location is far less important for planning academic and personal calendars than knowing the dates.
Because of this uncertainty, those of us who want to attend the meeting go through aggressive calendar-managing in order to avoid important conflicts. In what is now an annual two-month blockade, here are some typical approaches to events in February and March:
- Thinking about attending on the inpatient clinical service during those months? Bad idea.
- Want to go on a family vacation? Reconsider.
- Invited to speak in a medical school or postgraduate course, or to give medical grand rounds? Think again.
- Want to get married, or to adopt a puppy, or to sign up for that adult ed class on using power tools? Better wait until April.
When I try to explain this odd state of affairs to my non-ID colleagues, they are invariably perplexed, or say something naive like “Why don’t you check the conference website?”
That should be a good idea, so let’s do that. The conference website — retroconference.org — is in fact excellent, with a rapid search feature for abstracts, posters, and many terrific web casts.
That is, it was excellent — the site no longer works. In its place, we have this:
For the record, I have long assumed that we don’t know when CROI will be because the conference organizers don’t know yet either. (When I directly ask them, this is what they say — “We don’t know yet.”)
But if someone does know, and they’re hiding it — as in this example or this one — perhaps Edward Snowden can leak it when he lands in Bolivia, or Venezuela, or wherever he ends up.
July 3rd, 2013
First Year ID Fellows — What Do They Learn, and What Do They Hate?
In the weird calendar of academic medical centers, July 1 is the “official” first day of school.
In our ID program, however, we shifted it to July 5 a few years ago to avoid the interruption of the July 4 holiday at the beginning of the year. On July 3 — today — our incoming first-year fellows round with our ID fellows who have completed 363 days, acting as observers.
If ever you wanted tangible evidence that the first-year learning curve has a very large area under it, this is the day. Because the soon-to-be second years know a ton, the newbies … well, let’s just say there’s a reason they do an ID fellowship, and it’s to learn clinical ID!
In addition, fellows who have spent a year doing ID consults nearly every day also have learned a few things that they truly hate.
Here’s one: A late afternoon consult.
(That was easy.)
But rather than my providing the list, I would love to hear from you — two targeted questions:
- What do first-year fellows learn?
- What do they learn to hate?
Responses — anonymous and otherwise — welcome! And here’s a video:
June 27th, 2013
Testing Out the New Website with an ID Link-o-Rama
Hey, new website is live! Interested to hear what you think about our new-ish look.
In celebration, here are some quick ID/HIV tidbits that have recently crossed my path, or have been sitting in my inbox for a while, dying to get out:
- Doxycycline shortage. Hardly anything more frightening to a New England ID doc than a shortage of the world’s most versatile antibiotic during peak Lyme season. Shudder. Fortunately, most of my patients have had no trouble getting it from their pharmacies, though I do hear prices are way up.
- Some HCV news — USPSTF recommends screening baby-boomers (joining CDC, they had already recommended the same), and an HCV genotype test is approved. Here’s the question — why is the latter news at all? We’ve been sending HCV genotypes — “unapproved,” I guess — for years and years.
- More HCV: Did you see the CDC study published this Spring showing that around half the people with HCV don’t get a complete evaluation? Specifically, after a positive antibody test, many have no subsequent HCV RNA (viral load) checked to determine whether they have chronic disease. How is that possible?
- Another ID competition from John Bartlett. This time, it’s “Most Important Antiviral”. Go over to Medscape and vote and, more importantly, go read John’s fascinating discussion — which I bet he dictated between 4 and 5 a.m. early one day without using notes.
- A retrospective study says you can stop antibiotics on patients with ventilator-associated pneumonia if sputum cultures are negative. Yeah — RIGHT. (Readers of a certain age will say that with the full Bill Cosby effect.) NEJM Journal Watch summary here.
- Nice balanced piece in the New Yorker on the Lyme Disease controversy. Kudos to the author for not implying that there are villains on either side — a common mistake when the issue is covered in the lay press.
- And on the subject of ticks, this time of the Lone Star variety, how about this bizarre connection — tick bites and allergy to red meat. Note that the allergic reaction is delayed, which may make the connection difficult to diagnose. The article includes the inevitable advice about wearing long pants and tucking them into your socks during the summer. Again — RIGHT. Perhaps no public health advice is less heeded than that one, beating out even condom use. (Love the interview at the start of that video, by the way — a young couple doing condom testing for “science”!)
- The Kaiser Family Foundation has released this fascinating report on the funding sources for global health related to HIV/AIDS. Lots of interesting data, including the fact that the U.S. provides almost two thirds of all HIV/AIDS international assistance (61%), with the next largest donor, the Global Fund, providing one fifth — that’s around 80% of the total, if I’ve done my math right.
- Wouldn’t it be nice to have something that “eliminates up to 99.9% of airborne bacteria, mold, viruses”? And now, for just $299.95, it can be yours. Good thing it isn’t $300.00, that would be way too expensive.
By the way, wondering how to say “NEJM Group”? It’s N-E-J-M Group, spelled out, and not preceded by the word “The.” Now we all know. And I guess the Mass Medical Society prefers their doctors to be photographed wearing a tie.
Enjoy this classic comedy!
June 22nd, 2013
NEJM Group, Blog Freeze, and Puppy Brothers
It’s official — the various medical products of the Massachusetts Medical Society are now lumped under NEJM Group — including the flagship New England Journal of Medicine itself, the Journal Watches, even this site.
No surprise, therefore, that there will be a new web platform, with the launch slated for next week. I’ve been told this means a “blog freeze” — sounds like a summer ice cream treat — for at least June 24-25. During the freeze, there will be no new posts or comments.
Here then, to bide the time until we’re up and running again, is a picture of our puppy Louie and his brother Arlo, who fortunately just happens to live down the street:
June 20th, 2013
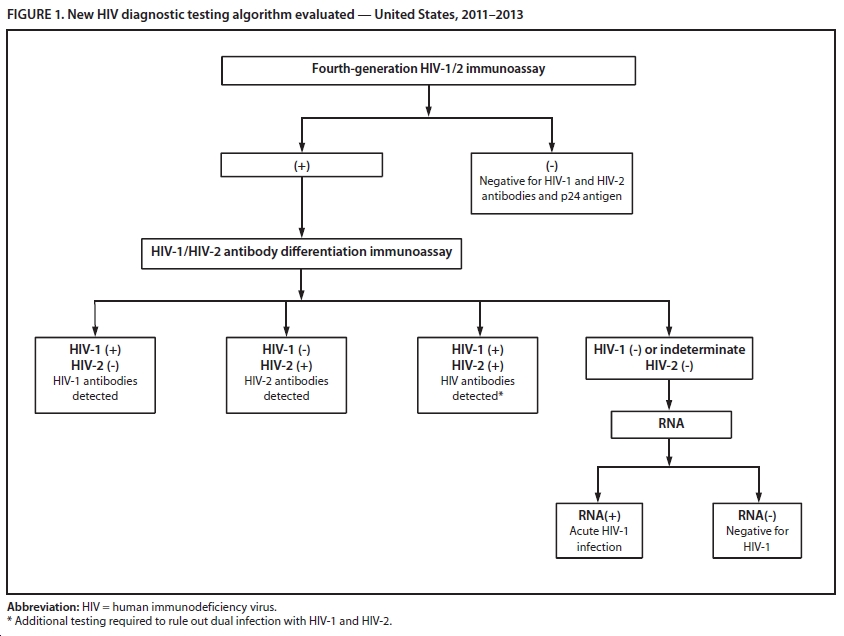
Let’s Move the HIV Testing Algorithm Into the 21st Century
As I’ve written before, the most widely used testing algorithm for HIV — enzyme immunoassay followed, if positive, by Western blot confirmation — is long overdue for an update.
A brief review why this is the case, and also why sticking with it is so problematic:
- Immunoassays have become progressively more sensitive, especially when paired with p24 antigen testing in “4th generation” tests.
- In recently acquired infection, the screening test turns positive way before the Western blot — it can be a difference of several weeks (figure from Branson J Acquir Immune Defic Syndr 2010;55:S102–S105):

- Clinicians are not familiar with this problem, so they might conclude that a reactive screening test followed by a negative Western blot means that the person doesn’t have HIV — it’s a “confirmatory test”, after all, so you can understand their confusion.
- Patients during this phase of early/acute HIV infection are at their most contagious, hence most likely to spread the virus to others — especially if given the wrong information about their HIV status.
- Patients may seek testing soon after high-risk exposures — they’re motivated! — and hence are potentially in this window before the Western blot turns positive.
The frustrating thing about this state of affairs is that we have had the tools to correct this problem for some time. Laboratory guidelines for HIV testing were updated in 2011, recommending the following algorithm for HIV diagnosis:
Now, thanks to CDC, we have some concrete data about how useful this new testing strategy can be — in particular for those with recently acquired HIV. A screening program in an Arizona emergency department identified 37 individuals with undiagnosed with HIV, and twelve of them — nearly a third — had acute HIV that otherwise would have been missed by the Western blot.
A second validation study looked at over 37,000 samples drawn from high-risk patients in sites from New York City, North Carolina, and San Francisco; there were 99 cases where the screening immunoassay was positive and the second antibody test negative. In this group, 55 had acute HIV infection (diagnosed by HIV RNA), many of whom had negative Western blots.
According to Bernie Branson — HIV testing guru from CDC — the FDA approval of the differentiation assay (step 2 in the algorithm) has led many labs to adopt the new strategy:
The number of labs doing this has picked up considerably since Multispot received its new indication … nearly half of public health labs (e.g., FL, MA, IL, IA, NY) have adopted it, as have Stroger/Cook County in Chicago, Howard University Hospital in DC, and LSU hospital system (to name a few). California has proposed emergency regulations (expected to be effective July 2) that will permit all labs in the state to perform the new algorithm, which their current regulation precludes, and New York State has issued 2 public health advisories encouraging its use.
It’s time for the rest of us to do the same.
And if you’re feeling bad for the Western blot, it still has a (rare) indication — as a confirmation of HIV infection in “elite controllers”, those with HIV antibodies but no detectable HIV RNA.
June 19th, 2013
FDA, IND, FMT: Nine Letters, Some Common Sense, and a Real Video Link
Good news here — the FDA has reconsidered their requirement for an IND for fecal microbiota transplantation (FMT) for C diff:
Some health care providers have stated that applying IND requirements will make FMT unavailable and have suggested that an alternative regulatory approach is needed to ensure the widespread availability of FMT for individuals with C. difficile infection unresponsive to standard therapies. The agency acknowledges these concerns and intends to exercise enforcement discretion regarding the IND requirements for the use of FMT to treat C. difficile infection not responding to standard therapies provided the treating physician obtains adequate informed consent from the patient.
This approach certainly makes sense.
Still, on reading the statement, one might wonder about the phrase “exercise enforcement discretion”. What precisely does this mean?
If I had to guess, they will choose to go after the clinicians who are using FMT for indications where the evidence for benefit is much weaker (thus far) than for C diff — e$pecially tho$e who $eem to be offering FMT for the wrong rea$on$.
And FYI, there are LOTS of those (still unproven) indications out there, as any brief internet search will reveal — as one site notes, they include “Ulcerative Colitis, Crohns, IBS, Constipation, Autoimmune disorders, Obesity, Parkinsons, Multiple Sclerosis, Anxiety and Depression.”
Will FMT work for some of these? As the cliche (and grant proposals) note, “more research is needed…”
Meanwhile, some were disappointed that the video I linked last time this topic was reviewed was a fake — you wanted the real thing.
Ask and you shall receive.